根據電化學腐蝕原理,腐蝕過程中產生的電流大小可以代表腐蝕速率。由于陽極極化和陰極極化使腐蝕電池電位減小,從而降低腐蝕速率。產生陽極極化的主要原因是在腐蝕過程中,當溶液中有氧化劑時,在陽極表面產生了保護性的氧化膜,使金屬鈍化。其電位正移可達0.2~2V,可使腐蝕速率降低幾個數量級。
工業上廣泛應用的(de)(de)(de)(de)鐵、鉻、鎳、鈦(tai)及其合金(jin)的(de)(de)(de)(de)活(huo)化(hua)(hua)-鈍(dun)化(hua)(hua)曲(qu)(qu)線具有(you)特殊(shu)的(de)(de)(de)(de)形(xing)式(shi),它們的(de)(de)(de)(de)活(huo)化(hua)(hua)-鈍(dun)化(hua)(hua)轉(zhuan)變的(de)(de)(de)(de)陽(yang)極(ji)(ji)(ji)(ji)極(ji)(ji)(ji)(ji)化(hua)(hua)曲(qu)(qu)線如圖(tu)9.1所示。圖(tu)中(zhong)有(you)三個不同(tong)電(dian)(dian)(dian)(dian)化(hua)(hua)學行(xing)為區(qu)(qu)域:活(huo)化(hua)(hua)區(qu)(qu)A、鈍(dun)化(hua)(hua)區(qu)(qu)P和(he)過鈍(dun)化(hua)(hua)區(qu)(qu)T。由于(yu)極(ji)(ji)(ji)(ji)化(hua)(hua)的(de)(de)(de)(de)作用,隨著腐(fu)(fu)蝕(shi)(shi)電(dian)(dian)(dian)(dian)流(liu)(liu)強(qiang)度(du)(du)的(de)(de)(de)(de)增加(jia),陽(yang)極(ji)(ji)(ji)(ji)電(dian)(dian)(dian)(dian)位(wei)E。升高(gao),當陽(yang)極(ji)(ji)(ji)(ji)極(ji)(ji)(ji)(ji)化(hua)(hua)曲(qu)(qu)線達到最(zui)(zui)大(da)值(zhi),相應電(dian)(dian)(dian)(dian)極(ji)(ji)(ji)(ji)電(dian)(dian)(dian)(dian)位(wei)為Ep,電(dian)(dian)(dian)(dian)流(liu)(liu)強(qiang)度(du)(du)為Ip時(shi)(shi),產生了陽(yang)極(ji)(ji)(ji)(ji)鈍(dun)化(hua)(hua),陽(yang)極(ji)(ji)(ji)(ji)過程受到極(ji)(ji)(ji)(ji)大(da)障礙,此(ci)時(shi)(shi)電(dian)(dian)(dian)(dian)流(liu)(liu)強(qiang)度(du)(du)突然(ran)下降到最(zui)(zui)小(xiao)值(zhi)I最(zui)(zui)小(xiao),Ep稱為初始鈍(dun)化(hua)(hua)電(dian)(dian)(dian)(dian)位(wei),Ip稱為臨界電(dian)(dian)(dian)(dian)流(liu)(liu)強(qiang)度(du)(du)。在很寬的(de)(de)(de)(de)陽(yang)極(ji)(ji)(ji)(ji)電(dian)(dian)(dian)(dian)位(wei)范圍內(nei)極(ji)(ji)(ji)(ji)化(hua)(hua)時(shi)(shi),一直保持(chi)I最(zui)(zui)小(xiao)的(de)(de)(de)(de)腐(fu)(fu)蝕(shi)(shi)電(dian)(dian)(dian)(dian)流(liu)(liu)強(qiang)度(du)(du),此(ci)時(shi)(shi)腐(fu)(fu)蝕(shi)(shi)速率大(da)大(da)降低,陽(yang)極(ji)(ji)(ji)(ji)處于(yu)鈍(dun)化(hua)(hua)區(qu)(qu)P。
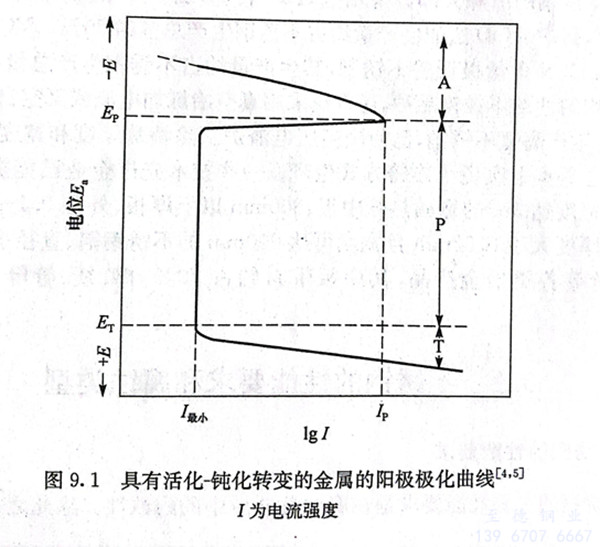
陽極(ji)電(dian)位超過(guo)(guo)Er后,腐蝕(shi)電(dian)流又增加(jia),這(zhe)種現象稱為(wei)過(guo)(guo)鈍化(hua)。ET稱為(wei)過(guo)(guo)鈍化(hua)電(dian)位,陽極(ji)處于過(guo)(guo)鈍化(hua)區(qu)T,此時(shi)金屬(shu)的腐蝕(shi)速率又增加(jia)。
根據具有活化(hua)-鈍(dun)化(hua)轉(zhuan)變的(de)金(jin)(jin)屬或合(he)金(jin)(jin)的(de)陽(yang)極(ji)(ji)極(ji)(ji)化(hua)曲(qu)線和(he)陰極(ji)(ji)極(ji)(ji)化(hua)曲(qu)線的(de)相對位置(zhi),可以分析該(gai)金(jin)(jin)屬和(he)合(he)金(jin)(jin)鈍(dun)化(hua)狀態的(de)穩定性(xing)。



