圖(tu)(tu)3.7(a)~(d)為(wei)(wei)鐵(tie)(tie)(tie)素體和(he)奧(ao)(ao)(ao)(ao)氏(shi)(shi)體相(xiang)(xiang)(xiang)中(zhong)的Cr、Mo、Ni元素含(han)(han)量(liang)(liang)(liang)(liang)(liang)(liang)(liang)與固溶(rong)溫(wen)(wen)度(du)的關(guan)系圖(tu)(tu)。圖(tu)(tu)3.7(a)~(d)證實(shi)了鐵(tie)(tie)(tie)素體相(xiang)(xiang)(xiang)中(zhong)的Cr和(he)Mo含(han)(han)量(liang)(liang)(liang)(liang)(liang)(liang)(liang)更高(gao)(gao),而奧(ao)(ao)(ao)(ao)氏(shi)(shi)體相(xiang)(xiang)(xiang)中(zhong)的Ni和(he)Mn含(han)(han)量(liang)(liang)(liang)(liang)(liang)(liang)(liang)更高(gao)(gao)。從圖(tu)(tu)中(zhong)可(ke)(ke)看出,鐵(tie)(tie)(tie)素體相(xiang)(xiang)(xiang)中(zhong)的Cr含(han)(han)量(liang)(liang)(liang)(liang)(liang)(liang)(liang)為(wei)(wei)23.77%~25.16%,比(bi)(bi)奧(ao)(ao)(ao)(ao)氏(shi)(shi)體相(xiang)(xiang)(xiang)中(zhong)Cr含(han)(han)量(liang)(liang)(liang)(liang)(liang)(liang)(liang)高(gao)(gao)2%左(zuo)右(you);鐵(tie)(tie)(tie)素體相(xiang)(xiang)(xiang)中(zhong)Mo含(han)(han)量(liang)(liang)(liang)(liang)(liang)(liang)(liang)為(wei)(wei)3.86%~4.37%,比(bi)(bi)奧(ao)(ao)(ao)(ao)氏(shi)(shi)體相(xiang)(xiang)(xiang)中(zhong)的Mo含(han)(han)量(liang)(liang)(liang)(liang)(liang)(liang)(liang)高(gao)(gao)1.7%左(zuo)右(you);奧(ao)(ao)(ao)(ao)氏(shi)(shi)體相(xiang)(xiang)(xiang)中(zhong)的Ni含(han)(han)量(liang)(liang)(liang)(liang)(liang)(liang)(liang)為(wei)(wei)5.42%、6.7%,比(bi)(bi)鐵(tie)(tie)(tie)素體相(xiang)(xiang)(xiang)中(zhong)的Ni含(han)(han)量(liang)(liang)(liang)(liang)(liang)(liang)(liang)高(gao)(gao)2%;當(dang)固溶(rong)處理(li)溫(wen)(wen)度(du)為(wei)(wei)1050℃時(shi),鐵(tie)(tie)(tie)素體相(xiang)(xiang)(xiang)中(zhong)的Cr、Mo和(he)Ni含(han)(han)量(liang)(liang)(liang)(liang)(liang)(liang)(liang)分別為(wei)(wei)23.77%、3.97%、4.24%,奧(ao)(ao)(ao)(ao)氏(shi)(shi)體相(xiang)(xiang)(xiang)中(zhong)的Cr、Mo和(he)Ni含(han)(han)量(liang)(liang)(liang)(liang)(liang)(liang)(liang)分別為(wei)(wei)23.53%、2.63%、5.42%.可(ke)(ke)見(jian)在1050℃溫(wen)(wen)度(du)下進(jin)行(xing)固溶(rong)時(shi),兩(liang)(liang)相(xiang)(xiang)(xiang)中(zhong)的Cr、Mo、Ni含(han)(han)量(liang)(liang)(liang)(liang)(liang)(liang)(liang)差(cha)(cha)異(yi)最小,此時(shi)鐵(tie)(tie)(tie)素體含(han)(han)量(liang)(liang)(liang)(liang)(liang)(liang)(liang)為(wei)(wei)51.9%,奧(ao)(ao)(ao)(ao)氏(shi)(shi)體含(han)(han)量(liang)(liang)(liang)(liang)(liang)(liang)(liang)為(wei)(wei)48.1%.當(dang)固溶(rong)溫(wen)(wen)度(du)改(gai)變(bian)時(shi),鐵(tie)(tie)(tie)素體/奧(ao)(ao)(ao)(ao)氏(shi)(shi)體兩(liang)(liang)相(xiang)(xiang)(xiang)比(bi)(bi)例相(xiang)(xiang)(xiang)差(cha)(cha)變(bian)大,且兩(liang)(liang)相(xiang)(xiang)(xiang)中(zhong)的Cr、Mo、Ni含(han)(han)量(liang)(liang)(liang)(liang)(liang)(liang)(liang)差(cha)(cha)異(yi)也變(bian)大。當(dang)固溶(rong)溫(wen)(wen)度(du)為(wei)(wei)1000℃,兩(liang)(liang)相(xiang)(xiang)(xiang)中(zhong)Cr為(wei)(wei)3.69%、Mo為(wei)(wei)1.51%、Ni為(wei)(wei)3.37%;當(dang)固溶(rong)溫(wen)(wen)度(du)為(wei)(wei)1150℃時(shi),兩(liang)(liang)相(xiang)(xiang)(xiang)中(zhong)Cr為(wei)(wei)2.29%、Mo為(wei)(wei)1.34%、Ni為(wei)(wei)2.09%,可(ke)(ke)見(jian)1000℃固溶(rong)試(shi)樣(yang)的兩(liang)(liang)相(xiang)(xiang)(xiang)中(zhong)的Cr、Mo、Ni元素含(han)(han)量(liang)(liang)(liang)(liang)(liang)(liang)(liang)差(cha)(cha)大于(yu)1150℃固溶(rong)試(shi)樣(yang)的兩(liang)(liang)相(xiang)(xiang)(xiang)中(zhong)Cr、Mo、Ni元素含(han)(han)量(liang)(liang)(liang)(liang)(liang)(liang)(liang)差(cha)(cha)。
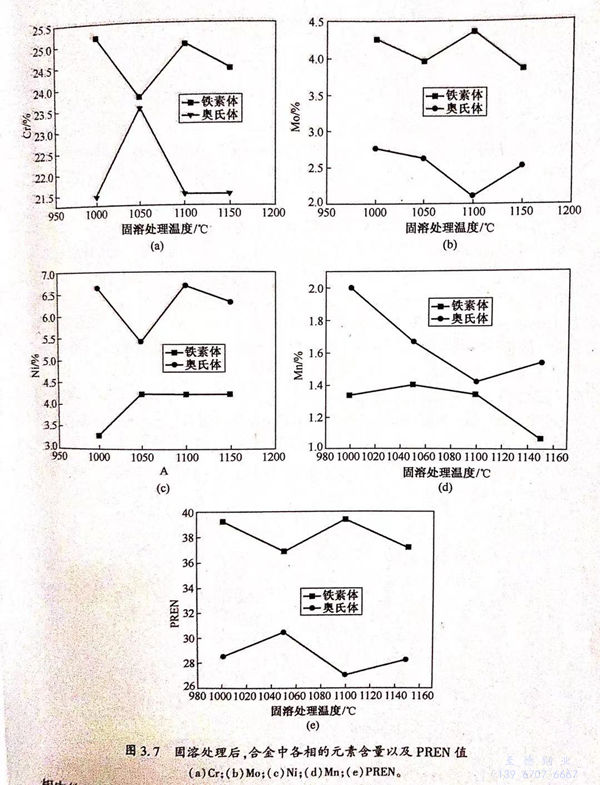
由公式“PREN.=[Cr]+3.3[Mo]”計算鐵素體(ti)的PREN值(zhi),由公式“PREN,=[Cr]+3.3[Mo]+16[N]-[Mn]”計算奧氏體(ti)的PREN值(zhi),可得PREN值(zhi)與固溶溫度(du)的關系圖,如圖3.7(e)所(suo)示。從(cong)圖3.7(e)可看(kan)出,在不同(tong)固溶狀態下,兩相(xiang)的PREN值(zhi)有(you)所(suo)不同(tong),但(dan)PRENa>PRENy.當固溶溫度(du)為(wei)(wei)1050℃時(shi),PREN.最(zui)小(xiao)、PREN,最(zui)大,分(fen)別為(wei)(wei)36.9和30.6,兩者相(xiang)差最(zui)小(xiao)。
圖(tu)3.8為不同(tong)固(gu)(gu)溶(rong)試樣的(de)極(ji)化曲線。可見,在自腐蝕(shi)電(dian)位下,材(cai)料開始發生鈍(dun)化;當(dang)極(ji)化電(dian)位升高到一(yi)定值時(shi),不同(tong)固(gu)(gu)溶(rong)的(de)材(cai)料都發生點(dian)蝕(shi),電(dian)流密度急(ji)劇增大。根據GB 4334.9-1984中(zhong)電(dian)流密度為0.1mA/c㎡所對應的(de)電(dian)位為點(dian)蝕(shi)電(dian)位和Tafel擬合,分別(bie)得到點(dian)蝕(shi)電(dian)位、自腐蝕(shi)電(dian)流密度與固(gu)(gu)溶(rong)溫(wen)度的(de)關系圖(tu),如圖(tu)3.11(a)所示(shi)。
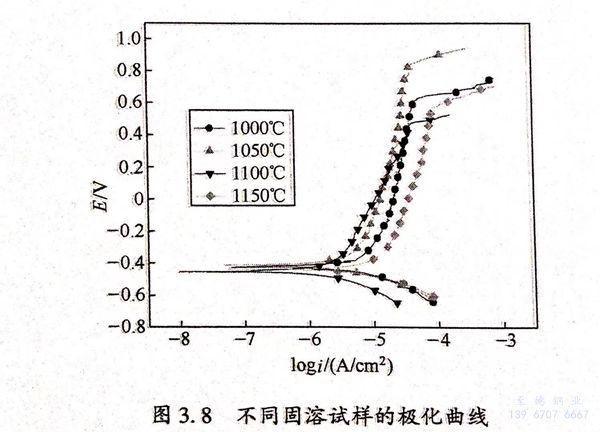
圖(tu)(tu)(tu)3.9(a)、(b)分(fen)別為不同(tong)固溶試(shi)樣(yang)的Nyquist 圖(tu)(tu)(tu)和(he)Bode圖(tu)(tu)(tu)。從圖(tu)(tu)(tu)3.9(a)可(ke)(ke)見(jian),試(shi)樣(yang)在(zai)低頻(pin)和(he)高(gao)頻(pin)區(qu)分(fen)別存(cun)(cun)在(zai)一個容(rong)抗(kang)弧(hu)。從圖(tu)(tu)(tu)3.9(b)可(ke)(ke)見(jian),試(shi)樣(yang)在(zai)低頻(pin)和(he)高(gao)頻(pin)處(chu)分(fen)別存(cun)(cun)在(zai)一個時間(jian)常(chang)數。根據(ju)曹楚南的《電(dian)(dian)化學(xue)阻(zu)抗(kang)譜(pu)導論》可(ke)(ke)知(zhi)。雙相不銹鋼在(zai)NaCl溶液(ye)中的阻(zu)抗(kang)譜(pu)中存(cun)(cun)在(zai)兩個時間(jian)常(chang)數,常(chang)以圖(tu)(tu)(tu)3.10所示(shi)(shi)的等效(xiao)電(dian)(dian)路(R1為電(dian)(dian)荷(he)(he)轉(zhuan)(zhuan)移電(dian)(dian)阻(zu),R2為鈍(dun)化膜電(dian)(dian)阻(zu))進(jin)行阻(zu)抗(kang)擬(ni)合。經阻(zu)抗(kang)擬(ni)合,得到鈍(dun)化膜電(dian)(dian)阻(zu)、電(dian)(dian)荷(he)(he)轉(zhuan)(zhuan)移電(dian)(dian)阻(zu)與固溶處(chu)理溫度(du)的關系圖(tu)(tu)(tu),如圖(tu)(tu)(tu)3.11(b)所示(shi)(shi)。
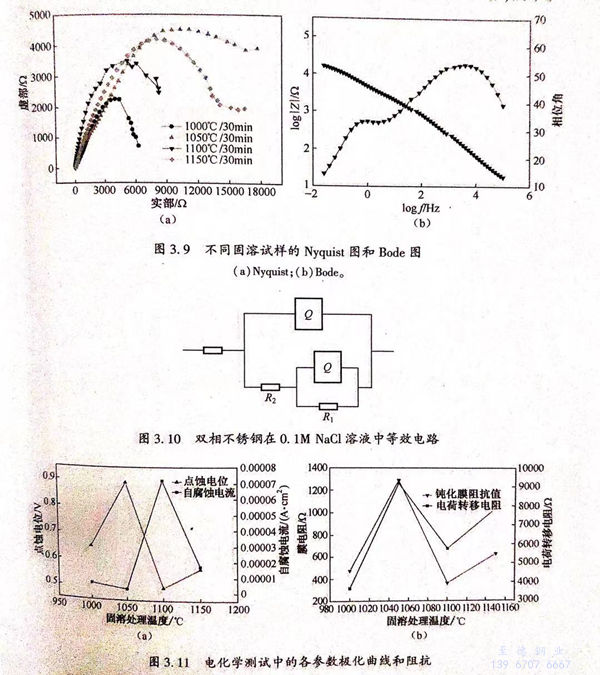
從圖3.11(a)可見,1050℃固溶試樣的點蝕電位最高,約為0.89V,且1050℃固溶試樣的自腐蝕電流密度最低,約為2.34×10-6A/c㎡,說明該狀態下試樣的耐點蝕性能最好。由圖3.11(b)可知,1050℃固溶試樣的鈍化膜電阻值最高,約為1300Ω,且其電荷轉移電阻值也最高,約為9500Ω,說明該狀態下試樣的鈍化膜較穩定,耐點蝕性能較好。
圖3.12為不同固溶的2205雙相不銹鋼極化后的點蝕形貌圖,圖中黑色為蝕坑,深色為鐵素體,淡色為奧氏體。從圖3.12可見,四種固溶試樣的點蝕都發生于鐵素體與奧氏體相界面處,且易向鐵素體相中長大。當固溶溫度為1050℃時,試樣的點蝕尺寸最小,點蝕敏感性最低。
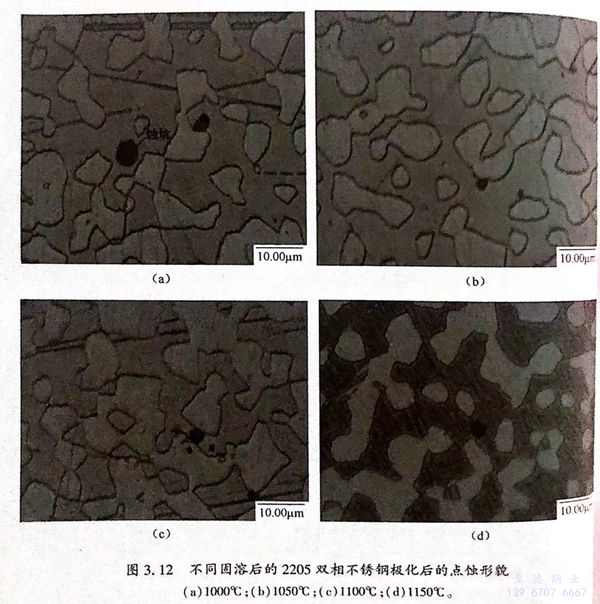
通(tong)過計算得到不(bu)同相比(bi)例下(xia)的(de)(de)(de)鐵素體PREN值(zhi)(zhi)和奧(ao)氏(shi)體PREN值(zhi)(zhi),可(ke)見不(bu)同相比(bi)例下(xia)的(de)(de)(de)PREN.都大于(yu)(yu)PREN,.雙(shuang)(shuang)相不(bu)銹鋼的(de)(de)(de)耐(nai)(nai)點(dian)(dian)蝕(shi)性(xing)能(neng)(neng)(neng)(neng)可(ke)通(tong)過點(dian)(dian)蝕(shi)電(dian)位(wei)衡量。點(dian)(dian)蝕(shi)電(dian)位(wei)越(yue)高,耐(nai)(nai)點(dian)(dian)獨性(xing)能(neng)(neng)(neng)(neng)越(yue)好(hao)。前人認為(wei)雙(shuang)(shuang)相不(bu)銹鋼的(de)(de)(de)耐(nai)(nai)點(dian)(dian)蝕(shi)性(xing)能(neng)(neng)(neng)(neng)由(you)B能(neng)(neng)(neng)(neng)N值(zhi)(zhi)較(jiao)區的(de)(de)(de)相決定,且PEN值(zhi)(zhi)越(yue)高,耐(nai)(nai)點(dian)(dian)蝕(shi)性(xing)能(neng)(neng)(neng)(neng)越(yue)好(hao),從(cong)圖3.71e)可(ke)矩,不(bu)同海(hai)溶試樣的(de)(de)(de)PREN.都大于(yu)(yu)PREN,,當固(gu)溶溫度為(wei)1050℃時,PHEN,最(zui)(zui)(zui)大,材料的(de)(de)(de)耐(nai)(nai)點(dian)(dian)蝕(shi)性(xing)能(neng)(neng)(neng)(neng)應最(zui)(zui)(zui)好(hao)。從(cong)圖3.11(a)、(b)可(ke)知(zhi),1050℃固(gu)溶試樣的(de)(de)(de)點(dian)(dian)蝕(shi)電(dian)位(wei)最(zui)(zui)(zui)高,鈍化膜阻(zu)抗值(zhi)(zhi)最(zui)(zui)(zui)大,電(dian)荷轉(zhuan)移電(dian)阻(zu)值(zhi)(zhi)最(zui)(zui)(zui)商;且從(cong)圖3.12(b)可(ke)見,1050℃固(gu)溶試樣的(de)(de)(de)點(dian)(dian)蝕(shi)坑尺(chi)寸最(zui)(zui)(zui)小,表現出最(zui)(zui)(zui)好(hao)的(de)(de)(de)耐(nai)(nai)點(dian)(dian)蝕(shi)性(xing)能(neng)(neng)(neng)(neng)。綜上可(ke)知(zhi),雙(shuang)(shuang)相不(bu)銹鋼的(de)(de)(de)耐(nai)(nai)點(dian)(dian)蝕(shi)性(xing)能(neng)(neng)(neng)(neng)由(you) PREN 值(zhi)(zhi)較(jiao)小相決定的(de)(de)(de)理(li)論(lun)是有實驗依據的(de)(de)(de)。
從(cong)圖3.12(a)~(d)可(ke)見,在不同(tong)固溶狀(zhuang)態下(xia),鐵(tie)素體相都更易(yi)發(fa)生點蝕;而從(cong)圖3.7(e)可(ke)見,在不同(tong)固溶狀(zhuang)態下(xia),PREN.都大于PREN,,鐵(tie)素體的耐(nai)點蝕性能應優(you)于奧氏體相,可(ke)見兩者存在矛盾。
隨(sui)固(gu)溶溫(wen)(wen)度(du)變(bian)化,雙(shuang)相(xiang)不(bu)(bu)(bu)銹鋼中(zhong)(zhong)(zhong)(zhong)(zhong)的(de)(de)(de)鐵素(su)(su)(su)(su)(su)(su)體和(he)奧(ao)氏(shi)(shi)體相(xiang)的(de)(de)(de)比例(li)改(gai)變(bian),且(qie)兩(liang)(liang)(liang)相(xiang)形(xing)態也(ye)發生(sheng)變(bian)化。Cr是鐵素(su)(su)(su)(su)(su)(su)體形(xing)成元(yuan)(yuan)(yuan)素(su)(su)(su)(su)(su)(su),可(ke)(ke)提(ti)高(gao)材料(liao)(liao)(liao)的(de)(de)(de)耐(nai)蝕(shi)(shi)(shi)(shi)性能;Mo是鐵素(su)(su)(su)(su)(su)(su)體形(xing)成元(yuan)(yuan)(yuan)素(su)(su)(su)(su)(su)(su),可(ke)(ke)提(ti)高(gao)點(dian)(dian)(dian)蝕(shi)(shi)(shi)(shi)電(dian)位,降低(di)腐蝕(shi)(shi)(shi)(shi)速率;Ni是奧(ao)氏(shi)(shi)體形(xing)成元(yuan)(yuan)(yuan)素(su)(su)(su)(su)(su)(su),可(ke)(ke)維持兩(liang)(liang)(liang)相(xiang)平衡,提(ti)高(gao)耐(nai)蝕(shi)(shi)(shi)(shi)性能。并且(qie)材料(liao)(liao)(liao)中(zhong)(zhong)(zhong)(zhong)(zhong)存(cun)在一定量(liang)的(de)(de)(de)N,其為奧(ao)氏(shi)(shi)體形(xing)成元(yuan)(yuan)(yuan)素(su)(su)(su)(su)(su)(su),提(ti)高(gao)局部腐蝕(shi)(shi)(shi)(shi)抗力(li)。從圖3.7(a)~(c)可(ke)(ke)見,隨(sui)固(gu)溶溫(wen)(wen)度(du)變(bian)化,兩(liang)(liang)(liang)相(xiang)中(zhong)(zhong)(zhong)(zhong)(zhong)的(de)(de)(de)Cr、Mo、Ni元(yuan)(yuan)(yuan)素(su)(su)(su)(su)(su)(su)含量(liang)發生(sheng)變(bian)化。由于(yu)Cr、Mo、Ni元(yuan)(yuan)(yuan)素(su)(su)(su)(su)(su)(su)之(zhi)間(jian)的(de)(de)(de)腐蝕(shi)(shi)(shi)(shi)電(dian)位存(cun)在差(cha)異,勢必造成兩(liang)(liang)(liang)相(xiang)之(zhi)間(jian)存(cun)在電(dian)化學差(cha)異,使腐蝕(shi)(shi)(shi)(shi)更(geng)易(yi)發生(sheng)。從圖3.7(e)可(ke)(ke)知,不(bu)(bu)(bu)同(tong)固(gu)溶狀態下材料(liao)(liao)(liao)的(de)(de)(de)PREN,大小關系(xi)為1050℃>1000℃>1150℃>1100℃,因(yin)此根據前人的(de)(de)(de)研究(jiu),不(bu)(bu)(bu)同(tong)固(gu)溶態材料(liao)(liao)(liao)的(de)(de)(de)耐(nai)點(dian)(dian)(dian)蝕(shi)(shi)(shi)(shi)性能優(you)劣關系(xi)應為:1050℃優(you)于(yu)1000℃優(you)于(yu)1150℃.當(dang)固(gu)溶溫(wen)(wen)度(du)為1050℃時,PREN,值較大,兩(liang)(liang)(liang)相(xiang)中(zhong)(zhong)(zhong)(zhong)(zhong)的(de)(de)(de)Cr、Mo、Ni元(yuan)(yuan)(yuan)素(su)(su)(su)(su)(su)(su)含量(liang)差(cha)異最(zui)小,材料(liao)(liao)(liao)的(de)(de)(de)點(dian)(dian)(dian)蝕(shi)(shi)(shi)(shi)坑較小,材料(liao)(liao)(liao)的(de)(de)(de)耐(nai)點(dian)(dian)(dian)蝕(shi)(shi)(shi)(shi)性能最(zui)優(you)。1000℃固(gu)溶試(shi)(shi)(shi)樣(yang)(yang)的(de)(de)(de)點(dian)(dian)(dian)蝕(shi)(shi)(shi)(shi)坑尺寸(cun)大于(yu)1150℃固(gu)溶試(shi)(shi)(shi)樣(yang)(yang)的(de)(de)(de)點(dian)(dian)(dian)蝕(shi)(shi)(shi)(shi)坑尺寸(cun),因(yin)此,前者的(de)(de)(de)耐(nai)點(dian)(dian)(dian)蝕(shi)(shi)(shi)(shi)性能劣于(yu)后者的(de)(de)(de)耐(nai)點(dian)(dian)(dian)蝕(shi)(shi)(shi)(shi)性能。由此可(ke)(ke)見,雙(shuang)相(xiang)不(bu)(bu)(bu)銹鋼中(zhong)(zhong)(zhong)(zhong)(zhong)的(de)(de)(de)相(xiang)腐蝕(shi)(shi)(shi)(shi)不(bu)(bu)(bu)能僅(jin)僅(jin)由PREN值來(lai)解釋。由圖3.7(a)~(c)可(ke)(ke)知,1000℃固(gu)溶試(shi)(shi)(shi)樣(yang)(yang)的(de)(de)(de)兩(liang)(liang)(liang)相(xiang)中(zhong)(zhong)(zhong)(zhong)(zhong)的(de)(de)(de)Cr、Mo、Ni元(yuan)(yuan)(yuan)素(su)(su)(su)(su)(su)(su)含量(liang)差(cha)大于(yu)1150℃固(gu)溶試(shi)(shi)(shi)樣(yang)(yang)的(de)(de)(de)兩(liang)(liang)(liang)相(xiang)中(zhong)(zhong)(zhong)(zhong)(zhong)的(de)(de)(de)Gr、Mo、Ni元(yuan)(yuan)(yuan)素(su)(su)(su)(su)(su)(su)含量(liang)差(cha),因(yin)此,雙(shuang)相(xiang)不(bu)(bu)(bu)銹鋼中(zhong)(zhong)(zhong)(zhong)(zhong)的(de)(de)(de)相(xiang)腐蝕(shi)(shi)(shi)(shi)還(huan)與兩(liang)(liang)(liang)相(xiang)中(zhong)(zhong)(zhong)(zhong)(zhong)的(de)(de)(de)元(yuan)(yuan)(yuan)素(su)(su)(su)(su)(su)(su)分布有關,并且(qie)還(huan)需考慮到(dao)材料(liao)(liao)(liao)中(zhong)(zhong)(zhong)(zhong)(zhong)點(dian)(dian)(dian)蝕(shi)(shi)(shi)(shi)敏感性較強(qiang)的(de)(de)(de)區(qu)域,如晶(jing)界(jie)和(he)相(xiang)界(jie)。
點(dian)蝕是一種局部腐蝕現象,是由氯離子破壞鈍化膜而導致的。點蝕產生后,蝕坑處的基體被暴露在溶液中,導致材料進一步發生腐蝕,蝕坑長大。蝕坑前長大速率由材料的均勻溶解速率決定,溶解越快,蝕坑長大速度越大。因此,雙相不銹鋼的耐點蝕性能由兩部分構成:控制點蝕萌生和控制蝕坑長大的能力。本書中的點蝕電位意味著點蝕已長大。影響點蝕萌生的因素不僅包括PREN 值,還包括兩相中的元素分布和兩相的比例。從本書的分析可見,點蝕的長大與元素分布有關,兩相中的元素分布越均勻,蝕坑的長大速度越慢。因此在1050℃固溶狀態下,兩相中的元素分布最均勻,PREN,最高,合金的點蝕電位最高,材料的耐點蝕性能最好。而1000℃固溶試樣兩相中的元素分布不均勻,導致點蝕電位較低,點蝕坑尺寸較大,降低材料的耐點蝕性能。
影(ying)響(xiang)(xiang)雙相(xiang)(xiang)不(bu)銹(xiu)鋼相(xiang)(xiang)腐蝕的(de)因(yin)素不(bu)僅包括(kuo)PREN值,還包括(kuo)各相(xiang)(xiang)中的(de)元素分布和兩相(xiang)(xiang)比例,并且三者互相(xiang)(xiang)影(ying)響(xiang)(xiang)、關系復雜。因(yin)此,不(bu)能僅憑PREN值的(de)大小來(lai)判斷(duan)耐點蝕能力(li),應該綜(zong)合考慮(lv)各因(yin)素的(de)影(ying)響(xiang)(xiang)。



