1. 動(dong)電位極化曲線分析
2507雙相不銹鋼經1000℃、1050℃、1100℃、1150℃及1200℃固溶處理后于30℃在3.5%NaCl溶液中的動電位極化曲線如圖5.3所示。從圖5.3可知各固溶溫度下2507雙相不銹(xiu)鋼的極化曲線形狀大體相同,都具有明顯的鈍化區域,且鈍化區域都較寬。國標GB 4334.9-1984規定陽極極化曲線上與電流密度值為100μA/c㎡相應的電位為擊破電位(Eb),擊破電位指金屬材料最先產生點腐蝕的電位,其值越高表明材料產生孔蝕的傾向越低。當2507雙相不(bu)銹鋼主要化學成分一定時,其表面保護膜的均勻性及穩定性跟鋼的兩相組織(α相及γ相)有著密切聯系,鋼中的α相和γ相分布均勻、相比例接近1:1時保護膜比較穩定且對鋼的點蝕行為有較好的阻礙作用;當鋼中的α相和γ相分布不均勻時易導致鋼表面保護膜遭受破壞,降低鋼的抗點蝕性能。經過不同固溶溫度處理的DSS2507的擊破電位(Eb)值如表5.1所列。從表5.1可知DSS2507于3.5%NaCl溶液中的擊破電位(Eb)隨著固溶熱處理溫度的上升呈現先變大后減小的走勢,1050℃時點蝕電位值最高,表明該固溶溫度下2507雙相鋼發生點蝕的傾向較低。固溶溫度為1000℃時2507雙相不銹鋼中鐵素體相跟奧氏體相界處有σ相析出,其附近較易形成貧鉻及貧鉬區域,造成該固溶熱處理溫度下鋼的抗點腐蝕能力較弱;1050℃時DSS2507中的兩相結構(鐵素體α相及奧氏體y相)分布較均勻,相比例約為1:1,有利于鋼表面鈍化膜的穩定,進而對點蝕具有較好的抑制作用,表現為點蝕電位Eb值較高;然而隨固溶熱處理溫度繼續上升,鐵素體α相量增多奧氏體γ相量減少,導致鐵素體形成元素Cr和Mo的濃度減小,而奧氏體形成元素N濃度升高,因此Cr、Mo和N在這兩種組織結構中的分布變得不均勻,由于Cr、Mo和N是鋼的主要耐點蝕元素,因此鋼的耐腐蝕能力降低。具體表現為點蝕電位E,值降低,所以在1050~1200℃溫度范圍內2507雙相不銹鋼的點蝕電位降低。此外從表5.1中列出的(Eb-Ecom)值可以看出其值變化趨勢跟點蝕電位(Eb)值的變化趨勢相同,都呈先升高后降低的趨勢。由于(Eb-Ecom)差值表示形核阻力的大小,因此由(Eb-Ecom)差值變化趨勢可知點蝕形核的阻力也是先增大后減小,即隨著固溶溫度的升高鋼的抗點蝕性能呈先增強后減弱的趨勢。此外從表5.1中可以看出,隨固溶熱處理溫度的上升自腐蝕電流密度(Icorr)先降低后增高,1050℃時自腐蝕電流密度較低為3.5367×10-7A/c㎡.綜上所述,固溶處理溫度為1050℃時2507雙相不銹鋼有較好的抗腐蝕能力。
固溶態為(wei)1050℃的(de)(de)2507雙相(xiang)(xiang)(xiang)不(bu)銹鋼于3.5%NaCl溶液中動電位極(ji)化后的(de)(de)腐蝕(shi)(shi)形(xing)貌(mao)如圖5.4所示,圖中深(shen)色是(shi)鐵(tie)(tie)素(su)體(ti)(ti)(ti)(ti)(ti)(ti)α相(xiang)(xiang)(xiang)、淺(qian)色是(shi)奧(ao)(ao)氏(shi)(shi)(shi)體(ti)(ti)(ti)(ti)(ti)(ti)y相(xiang)(xiang)(xiang)、黑色圓點(dian)(dian)是(shi)點(dian)(dian)蝕(shi)(shi)坑。從圖5.4可(ke)知點(dian)(dian)蝕(shi)(shi)坑主要存在于鐵(tie)(tie)素(su)體(ti)(ti)(ti)(ti)(ti)(ti)x相(xiang)(xiang)(xiang)上,這(zhe)說明奧(ao)(ao)氏(shi)(shi)(shi)體(ti)(ti)(ti)(ti)(ti)(ti)y相(xiang)(xiang)(xiang)比鐵(tie)(tie)素(su)體(ti)(ti)(ti)(ti)(ti)(ti)α相(xiang)(xiang)(xiang)要穩定(ding),有著較(jiao)(jiao)優的(de)(de)抗點(dian)(dian)腐蝕(shi)(shi)性(xing)能。奧(ao)(ao)氏(shi)(shi)(shi)體(ti)(ti)(ti)(ti)(ti)(ti)γ相(xiang)(xiang)(xiang)相(xiang)(xiang)(xiang)比于鐵(tie)(tie)素(su)體(ti)(ti)(ti)(ti)(ti)(ti)α相(xiang)(xiang)(xiang)有較(jiao)(jiao)好的(de)(de)抗點(dian)(dian)蝕(shi)(shi)性(xing)能的(de)(de)原因在于元(yuan)(yuan)(yuan)素(su)N的(de)(de)作用。元(yuan)(yuan)(yuan)素(su)N是(shi)奧(ao)(ao)氏(shi)(shi)(shi)體(ti)(ti)(ti)(ti)(ti)(ti)形(xing)成(cheng)元(yuan)(yuan)(yuan)素(su),于奧(ao)(ao)氏(shi)(shi)(shi)體(ti)(ti)(ti)(ti)(ti)(ti)γ相(xiang)(xiang)(xiang)中的(de)(de)溶解度要比在鐵(tie)(tie)素(su)體(ti)(ti)(ti)(ti)(ti)(ti)α相(xiang)(xiang)(xiang)中的(de)(de)大,此外N能夠讓Cr和Mo元(yuan)(yuan)(yuan)素(su)從鐵(tie)(tie)素(su)體(ti)(ti)(ti)(ti)(ti)(ti)α相(xiang)(xiang)(xiang)遷移(yi)到奧(ao)(ao)氏(shi)(shi)(shi)體(ti)(ti)(ti)(ti)(ti)(ti)γ相(xiang)(xiang)(xiang)中,N顯(xian)著影響著這(zhe)兩種元(yuan)(yuan)(yuan)素(su)在奧(ao)(ao)氏(shi)(shi)(shi)體(ti)(ti)(ti)(ti)(ti)(ti)γ相(xiang)(xiang)(xiang)和鐵(tie)(tie)素(su)體(ti)(ti)(ti)(ti)(ti)(ti)α相(xiang)(xiang)(xiang)中的(de)(de)分配,兩相(xiang)(xiang)(xiang)中合金元(yuan)(yuan)(yuan)素(su)含量的(de)(de)差值隨N元(yuan)(yuan)(yuan)素(su)含量的(de)(de)增多(duo)而降低(di),所以奧(ao)(ao)氏(shi)(shi)(shi)體(ti)(ti)(ti)(ti)(ti)(ti)γ相(xiang)(xiang)(xiang)的(de)(de)點(dian)(dian)蝕(shi)(shi)電位較(jiao)(jiao)鐵(tie)(tie)素(su)體(ti)(ti)(ti)(ti)(ti)(ti)α相(xiang)(xiang)(xiang)要高,從而蝕(shi)(shi)坑較(jiao)(jiao)先在Eb相(xiang)(xiang)(xiang)對比較(jiao)(jiao)低(di)的(de)(de)鐵(tie)(tie)素(su)體(ti)(ti)(ti)(ti)(ti)(ti)α相(xiang)(xiang)(xiang)中產(chan)生(sheng)。

2. 交流阻抗測(ce)試分析
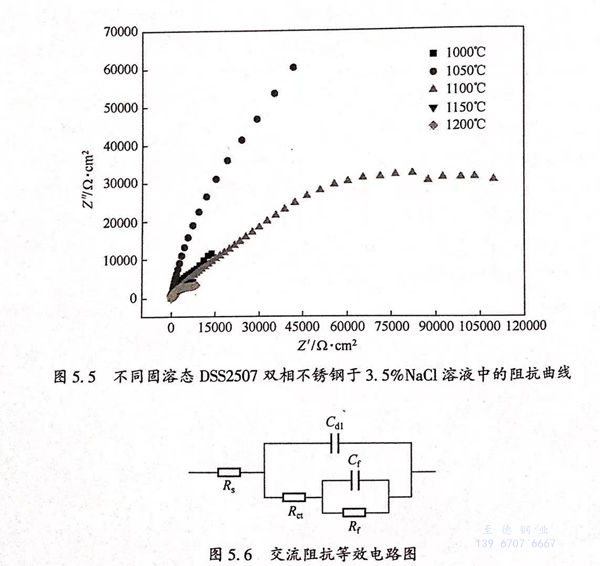
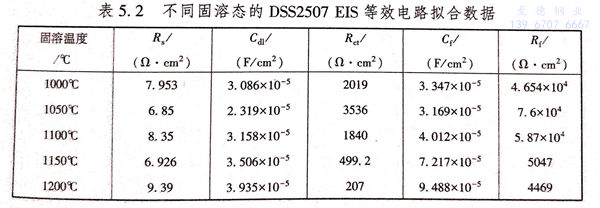
1000℃、1050℃、1100℃、1150℃及(ji)1200℃固溶(rong)(rong)(rong)態(tai)的(de)(de)(de)(de)(de)(de)2507 雙(shuang)(shuang)(shuang)相(xiang)(xiang)(xiang)不(bu)銹(xiu)(xiu)鋼(gang)(gang)30℃下于3.5%NaCl溶(rong)(rong)(rong)液(ye)(ye)中(zhong)的(de)(de)(de)(de)(de)(de)Nyquist 圖(tu)(tu)如圖(tu)(tu)5.5所(suo)示,從(cong)圖(tu)(tu)中(zhong)可(ke)以很明顯看出(chu)隨固溶(rong)(rong)(rong)熱處(chu)理溫(wen)(wen)度(du)(du)的(de)(de)(de)(de)(de)(de)增(zeng)高(gao)(gao)容(rong)(rong)(rong)抗(kang)(kang)(kang)(kang)弧半徑(jing)(jing)先(xian)增(zeng)大(da)后(hou)減(jian)(jian)小,1050℃時(shi)(shi)(shi)容(rong)(rong)(rong)抗(kang)(kang)(kang)(kang)弧半徑(jing)(jing)最(zui)大(da)。通常情(qing)況下界(jie)面間(jian)電(dian)(dian)荷(he)(he)(he)的(de)(de)(de)(de)(de)(de)遷(qian)(qian)(qian)移(yi)(yi)促進了高(gao)(gao)頻區容(rong)(rong)(rong)抗(kang)(kang)(kang)(kang)弧的(de)(de)(de)(de)(de)(de)形成,容(rong)(rong)(rong)抗(kang)(kang)(kang)(kang)弧與雙(shuang)(shuang)(shuang)電(dian)(dian)層容(rong)(rong)(rong)抗(kang)(kang)(kang)(kang)及(ji)金(jin)屬材(cai)料溶(rong)(rong)(rong)解(jie)的(de)(de)(de)(de)(de)(de)綜合作(zuo)用(yong)有(you)關,并且金(jin)屬-溶(rong)(rong)(rong)液(ye)(ye)界(jie)面的(de)(de)(de)(de)(de)(de)電(dian)(dian)荷(he)(he)(he)轉移(yi)(yi)電(dian)(dian)阻影響(xiang)著(zhu)(zhu)容(rong)(rong)(rong)抗(kang)(kang)(kang)(kang)弧半徑(jing)(jing)的(de)(de)(de)(de)(de)(de)大(da)小,電(dian)(dian)荷(he)(he)(he)在金(jin)屬-溶(rong)(rong)(rong)液(ye)(ye)界(jie)面轉移(yi)(yi)時(shi)(shi)(shi)受(shou)(shou)到的(de)(de)(de)(de)(de)(de)阻力(li)(li)越(yue)(yue)強(qiang)(qiang)意味著(zhu)(zhu)容(rong)(rong)(rong)抗(kang)(kang)(kang)(kang)弧半徑(jing)(jing)越(yue)(yue)大(da),即(ji)意味著(zhu)(zhu)金(jin)屬的(de)(de)(de)(de)(de)(de)抗(kang)(kang)(kang)(kang)蝕(shi)(shi)(shi)(shi)性能(neng)(neng)(neng)越(yue)(yue)優。從(cong)圖(tu)(tu)5.5容(rong)(rong)(rong)抗(kang)(kang)(kang)(kang)弧半徑(jing)(jing)的(de)(de)(de)(de)(de)(de)改變趨勢(shi)可(ke)知,固溶(rong)(rong)(rong)熱處(chu)理溫(wen)(wen)度(du)(du)升高(gao)(gao)使(shi)DSS2507的(de)(de)(de)(de)(de)(de)耐蝕(shi)(shi)(shi)(shi)性能(neng)(neng)(neng)首先(xian)增(zeng)強(qiang)(qiang)而后(hou)減(jian)(jian)弱,1050℃時(shi)(shi)(shi)2507雙(shuang)(shuang)(shuang)相(xiang)(xiang)(xiang)不(bu)銹(xiu)(xiu)鋼(gang)(gang)的(de)(de)(de)(de)(de)(de)抗(kang)(kang)(kang)(kang)腐(fu)(fu)蝕(shi)(shi)(shi)(shi)性能(neng)(neng)(neng)最(zui)優。不(bu)同固溶(rong)(rong)(rong)態(tai)的(de)(de)(de)(de)(de)(de)2507雙(shuang)(shuang)(shuang)相(xiang)(xiang)(xiang)不(bu)銹(xiu)(xiu)鋼(gang)(gang)于3.5%NaCl溶(rong)(rong)(rong)液(ye)(ye)中(zhong)的(de)(de)(de)(de)(de)(de)等(deng)效電(dian)(dian)路如圖(tu)(tu)5.6所(suo)示,采(cai)用(yong)的(de)(de)(de)(de)(de)(de)是R(C(R(CR))).Rs指參比電(dian)(dian)極和工作(zuo)電(dian)(dian)極間(jian)的(de)(de)(de)(de)(de)(de)溶(rong)(rong)(rong)液(ye)(ye)電(dian)(dian)阻;Cd1為(wei)(wei)雙(shuang)(shuang)(shuang)電(dian)(dian)層電(dian)(dian)容(rong)(rong)(rong);Rct為(wei)(wei)電(dian)(dian)荷(he)(he)(he)轉移(yi)(yi)電(dian)(dian)阻,其值能(neng)(neng)(neng)夠反應電(dian)(dian)荷(he)(he)(he)在電(dian)(dian)化(hua)(hua)學(xue)反應過(guo)程中(zhong)透(tou)過(guo)電(dian)(dian)極以及(ji)于電(dian)(dian)解(jie)質溶(rong)(rong)(rong)液(ye)(ye)中(zhong)兩(liang)相(xiang)(xiang)(xiang)界(jie)面遷(qian)(qian)(qian)移(yi)(yi)難易的(de)(de)(de)(de)(de)(de)大(da)小,也能(neng)(neng)(neng)夠反映金(jin)屬發(fa)生腐(fu)(fu)蝕(shi)(shi)(shi)(shi)反應的(de)(de)(de)(de)(de)(de)快慢,一(yi)般情(qing)況下Rct值越(yue)(yue)高(gao)(gao)意味著(zhu)(zhu)電(dian)(dian)荷(he)(he)(he)遷(qian)(qian)(qian)移(yi)(yi)過(guo)程中(zhong)所(suo)受(shou)(shou)的(de)(de)(de)(de)(de)(de)阻力(li)(li)越(yue)(yue)強(qiang)(qiang),即(ji)材(cai)料的(de)(de)(de)(de)(de)(de)耐腐(fu)(fu)蝕(shi)(shi)(shi)(shi)性能(neng)(neng)(neng)越(yue)(yue)好;Cf表(biao)(biao)示鈍(dun)(dun)化(hua)(hua)膜(mo)(mo)電(dian)(dian)容(rong)(rong)(rong),Rf表(biao)(biao)示鈍(dun)(dun)化(hua)(hua)膜(mo)(mo)電(dian)(dian)阻。采(cai)用(yong)ZsimpWin軟件(jian)對等(deng)效電(dian)(dian)路模擬(ni)計(ji)算得到各(ge)元件(jian)的(de)(de)(de)(de)(de)(de)數(shu)值如表(biao)(biao)5.2所(suo)列(lie)。從(cong)表(biao)(biao)中(zhong)看出(chu)雙(shuang)(shuang)(shuang)電(dian)(dian)層電(dian)(dian)容(rong)(rong)(rong)(Cdl)及(ji)鈍(dun)(dun)化(hua)(hua)膜(mo)(mo)電(dian)(dian)容(rong)(rong)(rong)(Cr)隨固溶(rong)(rong)(rong)溫(wen)(wen)度(du)(du)的(de)(de)(de)(de)(de)(de)升高(gao)(gao)呈(cheng)(cheng)先(xian)減(jian)(jian)小后(hou)增(zeng)大(da)的(de)(de)(de)(de)(de)(de)趨勢(shi);電(dian)(dian)荷(he)(he)(he)轉移(yi)(yi)電(dian)(dian)阻(Rct)和鈍(dun)(dun)化(hua)(hua)膜(mo)(mo)電(dian)(dian)阻(Rf)呈(cheng)(cheng)先(xian)變大(da)后(hou)減(jian)(jian)小的(de)(de)(de)(de)(de)(de)趨勢(shi),即(ji)電(dian)(dian)化(hua)(hua)學(xue)反應阻力(li)(li)隨固溶(rong)(rong)(rong)熱處(chu)理溫(wen)(wen)度(du)(du)的(de)(de)(de)(de)(de)(de)增(zeng)高(gao)(gao)先(xian)增(zeng)強(qiang)(qiang)后(hou)減(jian)(jian)弱,2507雙(shuang)(shuang)(shuang)相(xiang)(xiang)(xiang)不(bu)銹(xiu)(xiu)鋼(gang)(gang)的(de)(de)(de)(de)(de)(de)抗(kang)(kang)(kang)(kang)腐(fu)(fu)蝕(shi)(shi)(shi)(shi)性能(neng)(neng)(neng)表(biao)(biao)現為(wei)(wei)先(xian)增(zeng)強(qiang)(qiang)后(hou)減(jian)(jian)弱。1000℃時(shi)(shi)(shi)鋼(gang)(gang)的(de)(de)(de)(de)(de)(de)相(xiang)(xiang)(xiang)界(jie)處(chu)有(you)σ相(xiang)(xiang)(xiang)析出(chu),σ相(xiang)(xiang)(xiang)硬而脆降(jiang)低了鋼(gang)(gang)表(biao)(biao)面鈍(dun)(dun)化(hua)(hua)膜(mo)(mo)的(de)(de)(de)(de)(de)(de)穩(wen)定,表(biao)(biao)現為(wei)(wei)具(ju)有(you)較(jiao)(jiao)低的(de)(de)(de)(de)(de)(de)鈍(dun)(dun)化(hua)(hua)膜(mo)(mo)電(dian)(dian)阻(Rf);1050℃時(shi)(shi)(shi)鋼(gang)(gang)中(zhong)的(de)(de)(de)(de)(de)(de)兩(liang)相(xiang)(xiang)(xiang)比例約為(wei)(wei)1:1,兩(liang)相(xiang)(xiang)(xiang)分(fen)布(bu)較(jiao)(jiao)均勻且成分(fen)穩(wen)定有(you)利于鈍(dun)(dun)化(hua)(hua)膜(mo)(mo)的(de)(de)(de)(de)(de)(de)穩(wen)定,具(ju)體表(biao)(biao)現為(wei)(wei)鈍(dun)(dun)化(hua)(hua)膜(mo)(mo)電(dian)(dian)阻(Rf)值較(jiao)(jiao)高(gao)(gao),表(biao)(biao)明此刻(ke)離子遷(qian)(qian)(qian)移(yi)(yi)時(shi)(shi)(shi)所(suo)受(shou)(shou)阻力(li)(li)較(jiao)(jiao)大(da),進而2507雙(shuang)(shuang)(shuang)相(xiang)(xiang)(xiang)不(bu)銹(xiu)(xiu)鋼(gang)(gang)的(de)(de)(de)(de)(de)(de)抗(kang)(kang)(kang)(kang)腐(fu)(fu)蝕(shi)(shi)(shi)(shi)能(neng)(neng)(neng)力(li)(li)最(zui)佳;固溶(rong)(rong)(rong)熱處(chu)理溫(wen)(wen)度(du)(du)的(de)(de)(de)(de)(de)(de)進一(yi)步(bu)升高(gao)(gao)使(shi)鋼(gang)(gang)中(zhong)的(de)(de)(de)(de)(de)(de)兩(liang)相(xiang)(xiang)(xiang)比例失調,表(biao)(biao)現為(wei)(wei)鐵素體相(xiang)(xiang)(xiang)含量大(da)幅(fu)升高(gao)(gao),奧氏體相(xiang)(xiang)(xiang)含量減(jian)(jian)小,不(bu)利于鋼(gang)(gang)表(biao)(biao)面鈍(dun)(dun)化(hua)(hua)膜(mo)(mo)的(de)(de)(de)(de)(de)(de)穩(wen)定,導致鈍(dun)(dun)化(hua)(hua)膜(mo)(mo)電(dian)(dian)阻(Rf)減(jian)(jian)小,實際表(biao)(biao)現為(wei)(wei)鋼(gang)(gang)的(de)(de)(de)(de)(de)(de)抗(kang)(kang)(kang)(kang)蝕(shi)(shi)(shi)(shi)性能(neng)(neng)(neng)下降(jiang),這(zhe)與動電(dian)(dian)位掃描獲得的(de)(de)(de)(de)(de)(de)結(jie)論相(xiang)(xiang)(xiang)同。